UVa 1586 Molar mass(分子量)
2014-12-17 11:46
543 查看
Description

An organic compound is any member of a large class of chemical compounds whose molecules contain carbon. The molar mass of an organic compound is the mass
of one mole of the organic compound. The molar mass of an organic compound can be computed from the standard atomic weights of the elements.
When an organic compound is given as a molecular formula, Dr. CHON wants to find its molar mass. A molecular formula, such as C3H4O3 ,
identifies each constituent element by its chemical symbol and indicates the number of atoms of each element found in each discrete molecule of that compound. If a molecule contains more than one atom of a particular element, this quantity is indicated using
a subscript after the chemical symbol.
In this problem, we assume that the molecular formula is represented by only four elements, `C' (Carbon), `H' (Hydrogen), `O' (Oxygen), and `N' (Nitrogen) without parentheses.
The following table shows that the standard atomic weights for `C', `H', `O', and `N'.
For example, the molar mass of a molecular formula C6H5OH is 94.108 g/mol which is computed by 6 × (12.01 g/mol) + 6 × (1.008
g/mol) + 1 × (16.00 g/mol).
Given a molecular formula, write a program to compute the molar mass of the formula.
the input. Each test case is given in a single line, which contains a molecular formula as a string. The chemical symbol is given by a capital letter and the length of the string is greater than 0 and less than 80. The quantity number n which
is represented after the chemical symbol would be omitted when the number is 1 (2
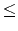
n
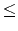
99) .
Sample
Sample
[/code]

An organic compound is any member of a large class of chemical compounds whose molecules contain carbon. The molar mass of an organic compound is the mass
of one mole of the organic compound. The molar mass of an organic compound can be computed from the standard atomic weights of the elements.

When an organic compound is given as a molecular formula, Dr. CHON wants to find its molar mass. A molecular formula, such as C3H4O3 ,
identifies each constituent element by its chemical symbol and indicates the number of atoms of each element found in each discrete molecule of that compound. If a molecule contains more than one atom of a particular element, this quantity is indicated using
a subscript after the chemical symbol.
In this problem, we assume that the molecular formula is represented by only four elements, `C' (Carbon), `H' (Hydrogen), `O' (Oxygen), and `N' (Nitrogen) without parentheses.
The following table shows that the standard atomic weights for `C', `H', `O', and `N'.
| Atomic Name | Carbon | Hydrogen | Oxygen | Nitrogen |
| Standard Atomic Weight | 12.01 g/mol | 1.008 g/mol | 16.00 g/mol | 14.01 g/mol |
g/mol) + 1 × (16.00 g/mol).
Given a molecular formula, write a program to compute the molar mass of the formula.
Input
Your program is to read from standard input. The input consists of T test cases. The number of test cases T is given in the first line ofthe input. Each test case is given in a single line, which contains a molecular formula as a string. The chemical symbol is given by a capital letter and the length of the string is greater than 0 and less than 80. The quantity number n which
is represented after the chemical symbol would be omitted when the number is 1 (2
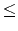
n
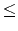
99) .
Output
Your program is to write to standard output. Print exactly one line for each test case. The line should contain the molar mass of the given molecular formula.Sample
Input
4 C C6H5OH NH2CH2COOH C12H22O11
Sample
Output
12.010 94.108 75.070 342.296
一道比较简单的字符串处理的题,根据题目给定的分子式的特点,只会有C,H,O,N四种,都是单个字符,所以不必考虑复杂化学式的处理,而给定的下标范围又是99以内,不会超过两位数,所以遍历一下字符串,当碰到大写字母时就判断后面两个字符的情况,根据是为一个数字还是两个数字还是没有数字来判断当前原子的个数,很容易累加得到结果。代码如下:
#include <iostream>
#include <cstring>
#include <iomanip>
using namespace std;
int main()
{
int word[128];//用来统计对应原子出现的次数
char str[111];
int n,len,i;
double res;
cin>>n;
while(n--)
{
memset(word,0,sizeof(word));//每组循环将数组清空
cin>>str;
len=strlen(str);
for(i=0;i<len;i++)
if(isupper(str[i]))
word[str[i]]+=(isdigit(str[i+1])?(isdigit(str[i+2])?((str[i+1]-48)*10+(str[i+2]-48)):str[i+1]-48):1);//通过判断原子后面的字符有几个是数字来决定统计的数量
res=word['C']*12.01+word['H']*1.008+word['O']*16.0+word['N']*14.01;//得出结果
cout<<setiosflags(ios::fixed)<<setprecision(3)<<res<<endl;
}
return 0;
}
[/code]
相关文章推荐
- 分子量(Molar Mass, ACM/ICPC Seoul 2007, UVa1586)
- UVA 1586 分子量(Molar Mass)
- UVa 1586 Molar mass (计算分子量,不含扩号)
- uva 1586 - Molar mass(分子量)
- 分子量 (Molar Mass,ACM/ICPC Seoul 2007,UVa 1586)
- 算法竞赛入门经典-习题3-2 分子量(Molar Mass, ACM/ICPC Seoul 2007, UVa1586)
- Molar Mass, ACM/ICPC Seoul 2007,Uva1586 给出一种物质的分子式,求分子量
- 算法竞赛入门竞赛 习题3-2 分子量(Molar Mass, ACM/ICPC Seoul 2007, UVa1586)
- UVa1586 Molar mass 分子量 (java版本)
- UVa1586-Molar Mass(分子量)
- UVa 1586 Molar Mass(分子量)
- 分子量 (Molar Mass, ACM/ICPC Seoul 2007, UVa1586)
- b2bf 分子量(Molar Mass, ACM/ICPC Seoul 2007, UVa1586)
- 习题3-2 分子量(Molar Mass, ACM/ICPC Seoul 2007, UVa1586)
- 小紫书 习题 3-2(UVA 1586) 分子量(Molar mass)
- 分子量(Molar Mass,UVa 1586)水题
- Uva1586-Molar Mass-分子量
- UVa1586 - Molar mass还是画流程图思路清晰
- UVa 1586 / UVALive 3900 Molar mass (字符串)
- 【习题 3-2 UVA - 1586】Molar mass
