Uva 1586 分子量
2014-11-01 16:58
183 查看
1586 - Molar mass
Time limit: 3.000 secondsAn organic compound is any member of a large class of chemical compounds whose molecules contain carbon. The molar mass of an organic compound is the mass
of one mole of the organic compound. The molar mass of an organic compound can be computed from the standard atomic weights of the elements.
When an organic compound is given as a molecular formula, Dr. CHON wants to find its molar mass. A molecular formula, such as C3 H4 O3 ,
identifies each constituent element by its chemical symbol and indicates the number of atoms of each element found in each discrete molecule of that compound. If a molecule contains more than one atom of a particular element, this quantity is indicated using
a subscript after the chemical symbol.
In this problem, we assume that the molecular formula is represented by only four elements, `C' (Carbon), `H' (Hydrogen), `O' (Oxygen), and `N' (Nitrogen) without parentheses.
The following table shows that the standard atomic weights for `C', `H', `O', and `N'.
| Atomic Name | Carbon | Hydrogen | Oxygen | Nitrogen |
| Standard Atomic Weight | 12.01 g/mol | 1.008 g/mol | 16.00 g/mol | 14.01 g/mol |
g/mol) + 1 × (16.00 g/mol).
Given a molecular formula, write a program to compute the molar mass of the formula.
Input
Your program is to read from standard input. The input consists of T test cases. The number of test cases T is given in the first line of theinput. Each test case is given in a single line, which contains a molecular formula as a string. The chemical symbol is given by a capital letter and the length of the string is greater than 0 and less than 80. The quantity number n which
is represented after the chemical symbol would be omitted when the number is 1 (2
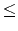
n
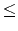
99) .
Output
Your program is to write to standard output. Print exactly one line for each test case. The line should contain the molar mass of the given molecular formula.Sample Input
4 C C6H5OH NH2CH2COOH C12H22O11
Sample Output
12.010 94.108 75.070 342.296
1、建立字母到数值的映射数组
2、碰到字母直接加原子量,碰到数字用(数字-1)乘以last atom的原子量
3、处理两位数,注意让i直接向后跳一步
#include <stdio.h>
#include <ctype.h>
char s[85];
const double d[] = {0,0,12.01,0,0,0,0,1.008,0,0,0,0,0,14.01,16.00};
int main(int argc, const char * argv[]) {
int T;
scanf("%d",&T);
while (T--) {
double sum = 0;
int n;
scanf("%s",s);
char last_atom = s[0];
for (int i = 0; s[i] != '\0'; ++ i) {
if (isalpha(s[i])) {
last_atom = s[i];
sum += d[last_atom-'A'];
}
else
{
n = s[i]-'0';
if(isnumber(s[i+1])){
n = (s[i]-'0')*10 + (s[i+1]-'0');
i ++;
}
sum += d[last_atom-'A']*(n-1);
}
}
printf("%.3f\n",sum);
}
return 0;
}
相关文章推荐
- 算法竞赛入门经典3.2 分子量UVa1586
- b2bf 分子量(Molar Mass, ACM/ICPC Seoul 2007, UVa1586)
- 分子量UVa1586
- UVa1586 Molar mass 分子量 (java版本)
- 水题 UVA 1586 - Ancient Cipher化学式分子量计算
- 分子量(UVa1586)
- 习题3-2 分子量(Molar Mass, ACM/ICPC Seoul 2007, UVa1586)
- 分子量 (Molar Mass, ACM/ICPC Seoul 2007, UVa1586)
- UVa1586-Molar Mass(分子量)
- UVa 1586 Molar Mass(分子量)
- 分子量(Molar Mass,UVa 1586)水题
- UVA 1586 分子量
- Uva1586-Molar Mass-分子量
- 习题3-2 分子量 UVa1586
- uva 1586 - Molar mass(分子量)
- UVA 1586 分子量(Molar Mass)
- 小紫书 习题 3-2(UVA 1586) 分子量(Molar mass)
- 算法竞赛入门竞赛 习题3-2 分子量(Molar Mass, ACM/ICPC Seoul 2007, UVa1586)
- UVa1586 分子量
- UVA1586 分子量 元素下标任意位数版本
